Biochemistry - The chemicals of life
Water
Molecular Structure of Water
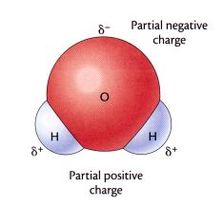
--Water is a polar molecule. --It consists of 1 oxygen atom and 2 hydrogen atoms covalently bonded. This means that the valence electrons are being shared. However, it is an uneven (or unequal) sharing. This is a type of covalent bond called polar covalent. It makes the oxygen side of the water molecule is slightly negative and the hydrogen side slightly positive (opposite poles). Hence , the reason why water is a polar molecule.
--Water is a polar molecule. --It consists of 1 oxygen atom and 2 hydrogen atoms covalently bonded. This means that the valence electrons are being shared. However, it is an uneven (or unequal) sharing. This is a type of covalent bond called polar covalent. It makes the oxygen side of the water molecule is slightly negative and the hydrogen side slightly positive (opposite poles). Hence , the reason why water is a polar molecule.
|
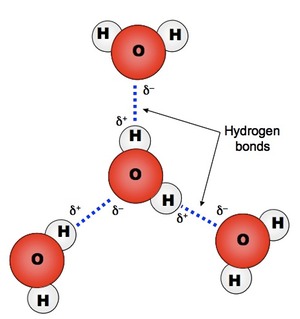
Hydrogen Bonding
Since a water molecule has a positive and negative side it acts almost like a tiny magnet. When many water molecules are together, they align so the hydrogen on one molecule will face the oxygen on another molecule. The bond that forms between two water molecules is a special bond called a hydrogen bond. It is a weak bond individually but many hydrogen bonds can have great intermolecular force. This gives water some very unique properties... |
The Unique Properties of Water
1. Universal Solvent: Because water is polar, it can dissolve many things very well.
2. Cohesion
3. Adhesion
4. Resists Changes in Temperature
High specific heat
High heat of vaporization
High heat of fusion
5. Expands upon freezing
becomes less dense when in ice form and will float in liquid water
1. Universal Solvent: Because water is polar, it can dissolve many things very well.
2. Cohesion
3. Adhesion
4. Resists Changes in Temperature
High specific heat
High heat of vaporization
High heat of fusion
5. Expands upon freezing
becomes less dense when in ice form and will float in liquid water
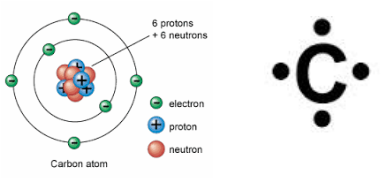
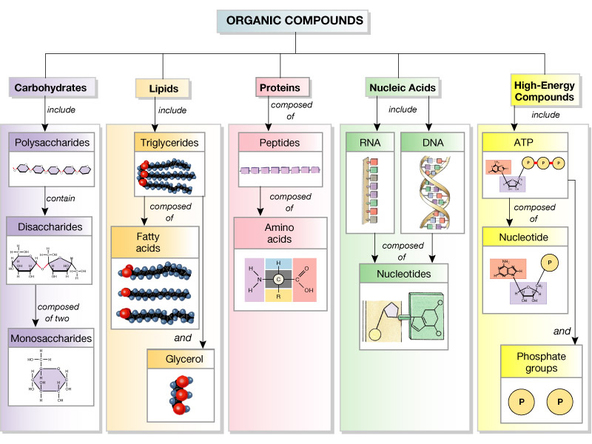
Carbon Compounds (organic chemistry)
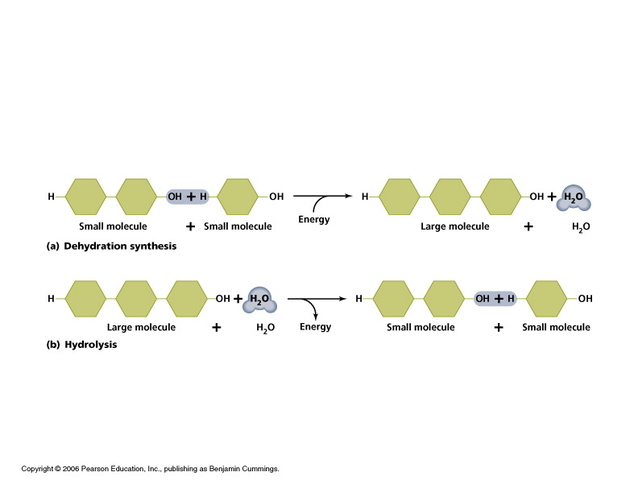
Polymers are formed by the bonding of monomers. The monomers join by a process called dehydration synthesis in which an enzyme removes the -OH from one monomer and a -H from another monomer. This forms a water molecule as a byproduct.
Polymers can be broken down in a process called hydrolysis which is the opposite of dehydration synthesis. Water is inserted to split the monomers apart.
Enzymes
What are Enzymes?
Enzymes are special types of proteins. Enzymes are responsible for a lot of the work that is going on in cells. They act as catalysts in order to help produce and speed up chemical reactions. When a cell needs to get something done, it almost always uses an enzyme to speed things along. The molecule or substance that the enzyme reacts with is called the "substrate."
Like all proteins, enzymes are made from chains of amino acids. These protein chains are folded into very precise shapes that create an active site - a place on the enzyme where the substrate attaches and the chemical reaction takes place. Changing the order of amino acids would cause it to fold into a different shape. Therefore, each enzyme's active site is suitable for one specific type of substrate – just like a lock that has the right shape for only one specific key.
Changing the shape of the active site of an enzyme will cause its reaction to slow down until the shape has changed so much that the substrate no longer fits. When this happens the reaction stops. At this point we say the enzyme is denatured and is permanently damaged.
Things that Affect Enzyme Activity
The environment of the enzyme and the substrate can affect the speed of the reaction. In some cases the environment can cause the enzyme to stop working or even unravel. When an enzyme stops working we call it "denatured." Here are some things that can affect enzyme activity:
Enzymes are special types of proteins. Enzymes are responsible for a lot of the work that is going on in cells. They act as catalysts in order to help produce and speed up chemical reactions. When a cell needs to get something done, it almost always uses an enzyme to speed things along. The molecule or substance that the enzyme reacts with is called the "substrate."
Like all proteins, enzymes are made from chains of amino acids. These protein chains are folded into very precise shapes that create an active site - a place on the enzyme where the substrate attaches and the chemical reaction takes place. Changing the order of amino acids would cause it to fold into a different shape. Therefore, each enzyme's active site is suitable for one specific type of substrate – just like a lock that has the right shape for only one specific key.
Changing the shape of the active site of an enzyme will cause its reaction to slow down until the shape has changed so much that the substrate no longer fits. When this happens the reaction stops. At this point we say the enzyme is denatured and is permanently damaged.
Things that Affect Enzyme Activity
The environment of the enzyme and the substrate can affect the speed of the reaction. In some cases the environment can cause the enzyme to stop working or even unravel. When an enzyme stops working we call it "denatured." Here are some things that can affect enzyme activity:
- Temperature - The temperature can affect the reaction rate. The higher the temperature, the faster the reaction will occur. However, at some point the temperature will become so high that the enzyme will denature and stop working.
- pH - In many cases the pH level, or acidity, of the environment around the enzyme and substrate can affect the reaction rate. An extreme pH (high or low) will typically slow the reaction or even stop the reaction altogether.
- Concentration - A higher concentration of substrate or enzyme can increase the reaction rate.
Videos and helpful websites
|
The Amoeba Sisters put the biomolecules in simple terms.
|
|
|
Mr. Andersen's videos about the macromolecules (AKA polymers). He's awesome!
|
|
|
Wath this video to learn about protein structure.
|
|
|
Here's the enzyme video shown in class. :)
|
|